 In that edition, botanicals were named first by the Latin genus and species and then with the common name in parenthesis (i.e. The goal, I think, was to standardize and harmonize the ingredient naming standards between the US and other countries, which were going toward requiring the Latin names for ingredients. Most people, including myself, figured that the PCPC and the FDA were on the same page and that, so long as the common name was included, any of the different versions were okay to use. It appears that when it comes to using the Latin name for botanicals in ingredient listings, the FDA and PCPC aren’t on the same page after all. In the last several months, the FDA posted several documents on their website that give some insight into their position. In March 1995, the CTFA wrote the FDA in preparation of their upcoming 6th edition of the Cosmetic Ingredient Dictionary, requesting that the FDA agree not to take action against labelers using the Latin genus and species to identify botanicals. 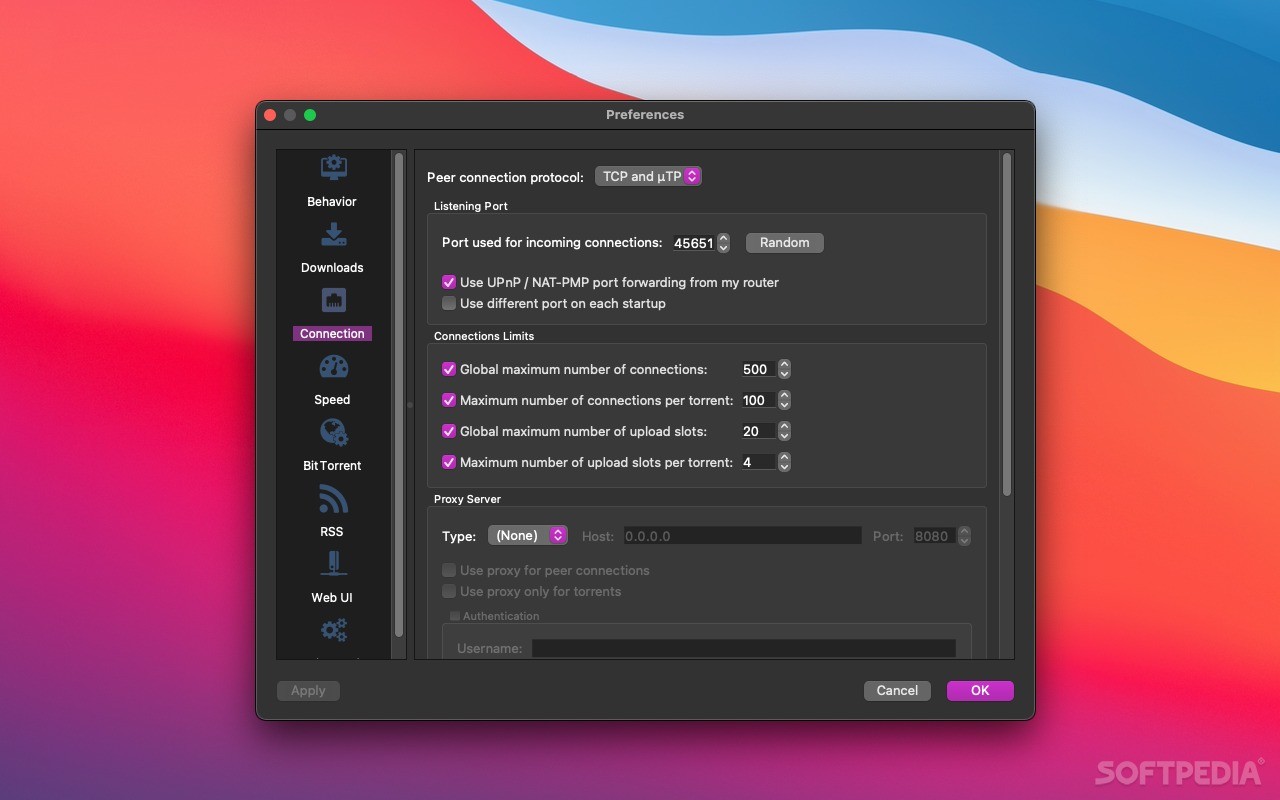
The section concerning the Latin names for botanicals is quoted here : They sent a follow-up letter changing the original request, asking that dual names be allowed as part of a “transition period”. 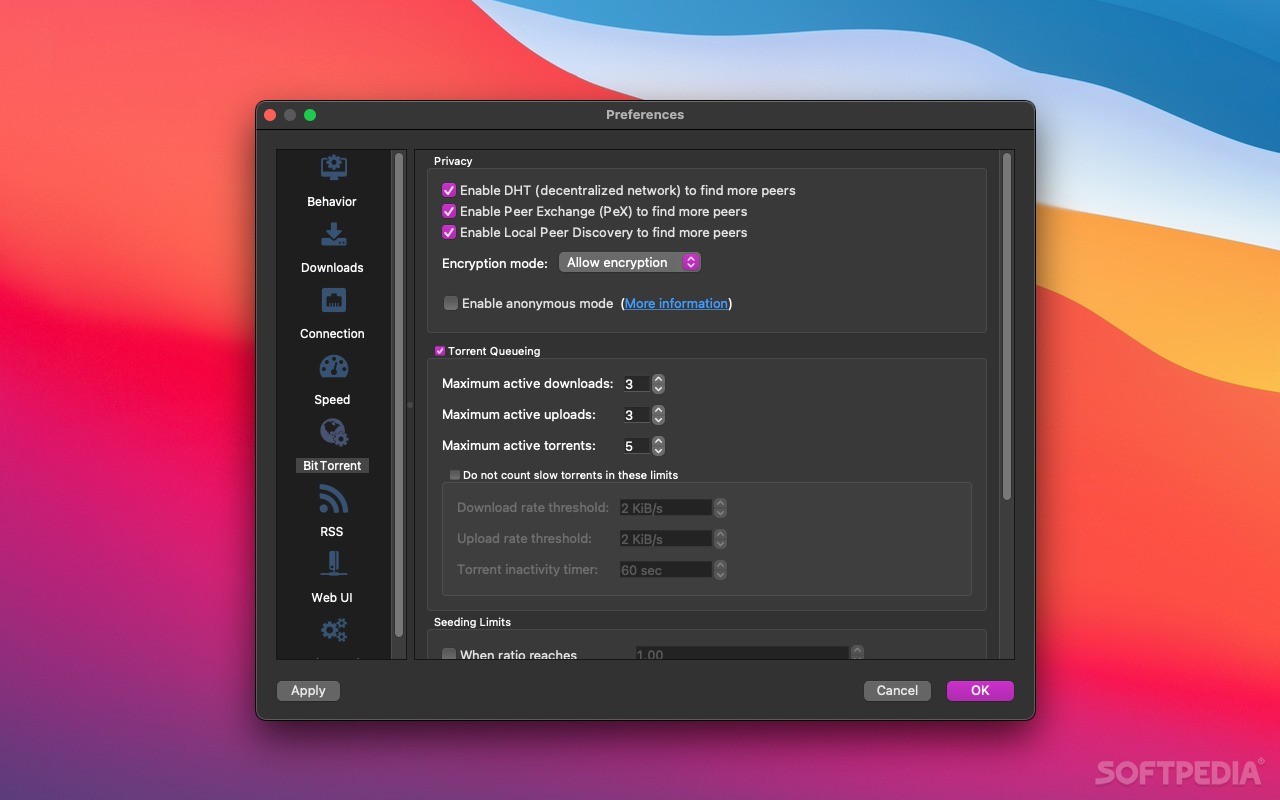
The use of Latin names as the primary identifying term for plant extract ingredients, with the current common name appearing imbedded in parenthesis, would not be consistent with the FPLA. 
The statute requires the use of the common or usual name, and there is no way that such a requirement can be considered to be met by the placement of the recognized common or usual name in parentheses after the Latin name. Later in the document, the FDA went on to say: Nor is the agency willing to accept the Latin name as the common or usual name of such ingredients. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
